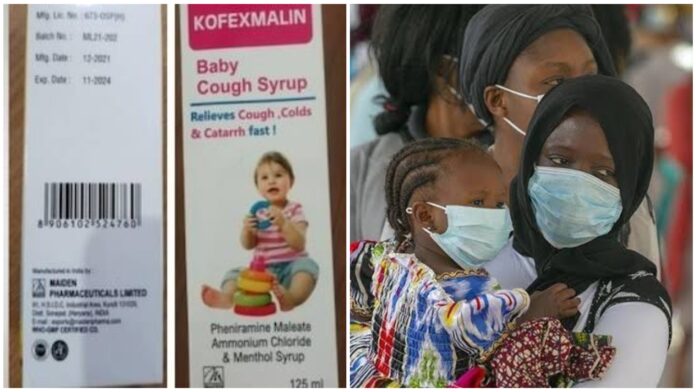
The death of dozens of children in The Gambia from kidney injuries could be linked to contaminated coughing and cold syrups from an Indian drugmaker, the World Health Organization said on Wednesday.
WHO Director-General Tedros Adhanom Ghebreyesus told reporters that the UN agency was investigating along with Indian regulators and New Delhi-based drugmaker Maiden Pharmaceuticals.
Maiden declined to comment on the warning, while calls and messages from Reuters to India’s Controller General of Drugs went unanswered. The Indian Ministry of Health also did not immediately respond to a request for comment.

The WHO has issued a medical device alert urging regulators to withdraw Maiden Pharmaceuticals products from the market. The products may have been distributed elsewhere through informal markets, but so far they have only been identified in The Gambia, the WHO said in its alert.
Also, read; Rwanda Ranked The Safest In Africa and 6th Safest Country For Solo Travelers
The warning was issued as regards four products: Promethazine Oral Solution, Kofexmalin Baby Cough Syrup, Makoff Baby Cough Syrup, and Magrip N Cold Syrup.
Laboratory analysis confirmed unacceptable levels of diethylene glycol and ethylene glycol, which can be toxic if ingested, the WHO said. The Gambian government said last month it was also investigating the deaths, following a spike in cases of acute kidney injury in children under five in late July.

Several children in The Gambia developed kidney problems three to five days after ingesting a locally sold acetaminophen syrup. In August, 28 died, but health officials said the number was likely to rise. Now 66 are dead, the WHO said on Wednesday.
The deaths have shaken the tiny West African nation, which is also facing multiple health emergencies, including measles and malaria.
Maiden Pharmaceuticals manufactures drugs at its facilities in India, which it then sells domestically and exports to countries in Asia, Africa and Latin America, according to its website.